COVID-19: Moderna announces it's first Coronavirus vaccine shows positive immune response in the U.S.
Moderna, the company, manufacturing a vaccine against the COVID-19 virus, on Monday announced that the first phase of testing the vaccine on people in the United States was able to stimulate an immune response against Coronavirus and appears to be safe. The results are of a vaccine test, which was c
Moderna,
the company, manufacturing a vaccine against the COVID-19 virus, on Monday
announced that the first phase of testing the vaccine on people in the United
States was able to stimulate an immune response against Coronavirus and appears
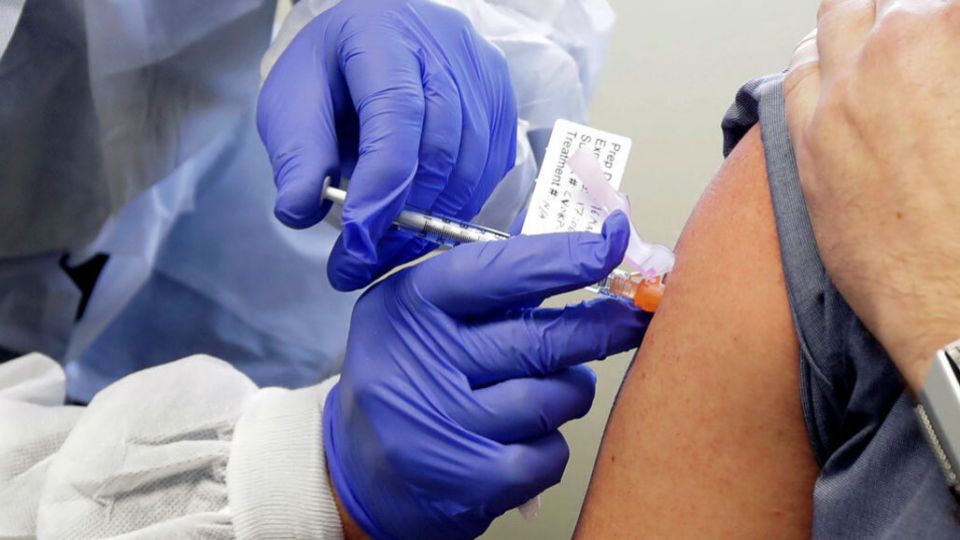
to be safe. The results are of a vaccine test, which was conducted on eight
people, were each given two doses of the vaccine since March. After the dose,
these healthy volunteers generated antibodies. At the company's laboratory, the
antibodies then were thoroughly tested in the human cells and resulted in
stopping the virus from multiplying.
The amount of the said neutralized
antibodies were at par with the amount found in the patients who had recovered
from the Coronavirus. The Vaccine was tested as low dose, medium dose and high
dose, low and medium doses had one side effect though, causing redness
and soreness, where the shot was given, in just one patient. The high dose
showed the best results; however further tests will be conducted on low and
medium doses as firm's chief medical officer Tai Zaks said, "Lower the
dose, more vaccine we'll be able to make". The second phase of the testing
which involves 600 people is due to start soon and the third phase will test
thousands of healthy people & is scheduled to begin from July said Moderna.
The FDA has given permission to
Moderna for further phases of testing. If all the testing goes well, then the
vaccine can be made available to the general public by 2020 end or early 2021.
Due to this positive announcement in pre-market trading, Moderna's stock was up
by 40%.
Disclaimer: The opinions expressed within this article are the personal opinions of the author. The facts and opinions appearing in the article do not reflect the views of 25hrNews and 25hrNews does not assume any responsibility or liability for the same.






 (+91) 8081113111
(+91) 8081113111 pr@25hrnews.com
pr@25hrnews.com